Qualityze Document Management Software is an AI-powered, cloud-based solution designed to help Life Sciences and Manufacturing organizations efficiently create, control, and manage quality documents while maintaining continuous compliance. Built natively on Salesforce, the platform provides real-time visibility, secure version control, and complete traceability across the entire document lifecycle.
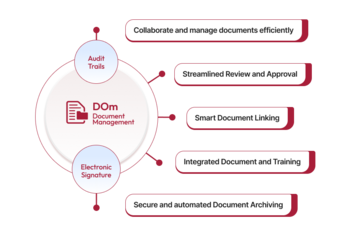
Qualityze enables quality and compliance teams to author, review, approve, distribute, and retire documents within a centralized system. By automating document workflows and enforcing standardized controls, organizations can eliminate manual errors, ensure employees always access the latest approved versions, and strengthen audit readiness.
The solution offers configurable approval workflows, role-based access, automated training linkage, and comprehensive reporting dashboards that provide clear insights into document status, revision history, and compliance gaps. This helps teams improve governance, enhance collaboration, and maintain consistent process control across the enterprise.
Purpose-built for regulated environments, Qualityze Document Management Software supports compliance with FDA, ISO 13485, and GxP requirements while enabling organizations to manage controlled documents with confidence and efficiency.