Qualityze Material Compliance Management Software is an AI-powered, cloud-based solution designed to help Life Sciences and Manufacturing organizations monitor and control material compliance across the supply chain while maintaining continuous regulatory adherence. Built natively on Salesforce, the platform provides real-time visibility, standardized workflows, and complete traceability for material qualification and compliance activities.
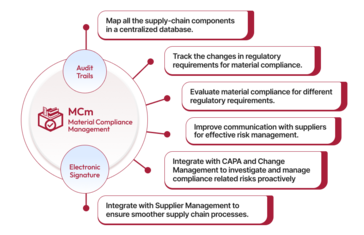
Qualityze enables quality, regulatory, and supply chain teams to track material specifications, manage compliance documentation, monitor supplier declarations, and ensure components meet required regulatory standards within a centralized system. By replacing manual tracking and fragmented tools, organizations can reduce compliance risk, improve material transparency, and strengthen product quality.
The solution offers configurable workflows, role-based access, automated notifications, and integrated reporting dashboards that deliver actionable insights into material status, compliance gaps, and supplier performance. This helps teams proactively manage regulatory obligations, improve cross-functional collaboration, and remain audit-ready.
Purpose-built for regulated environments, Qualityze Material Compliance Management Software supports compliance with FDA, ISO 13485, and GxP requirements while enabling organizations to maintain full visibility and control over material compliance.