CAPA Management Software
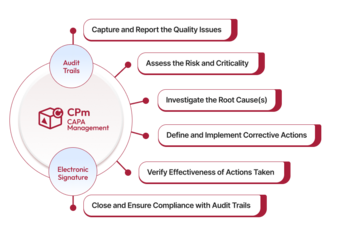
Qualityze CAPA Management Software is an AI-powered, cloud-based solution designed to help Life Sciences and Manufacturing organizations efficiently manage corrective and preventive actions while maintaining continuous compliance. Built natively on Salesforce, the platform provides real-time visibility, standardized workflows, and full traceability across the entire CAPA lifecycle. Qualityze enables quality teams to capture issues, perform root cause analysis, initiate corrective and preventive actions, and track effectiveness in a centralized system. By automating manual processes and enforcing structured workflows, organizations can accelerate investigations, reduce recurring issues, and improve overall product quality. The solution offers configurable workflows, risk-based prioritization, automated notifications, and comprehensive reporting dashboards that provide actionable insights into CAPA performance and trends. This helps teams strengthen quality processes, improve accountability, and stay audit-ready at all times. Purpose-built for regulated environments, Qualityze CAPA Management Software supports compliance with FDA, ISO 13485, and GxP requirements while driving continuous improvement and operational excellence.
When users leave CAPA Management Software reviews, G2 also collects common questions about the day-to-day use of CAPA Management Software. These questions are then answered by our community of 850k professionals. Submit your question below and join in on the G2 Discussion.
0.0
Nps Score
All CAPA Management Software Discussions
Start a New Software Discussion
Have a software question?
Get answers from real users and experts
Start A Discussion